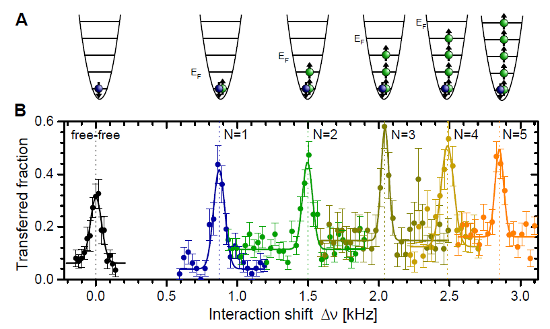
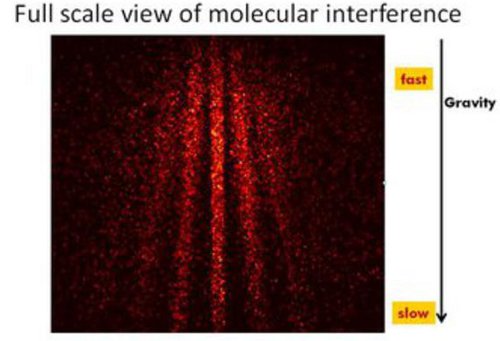
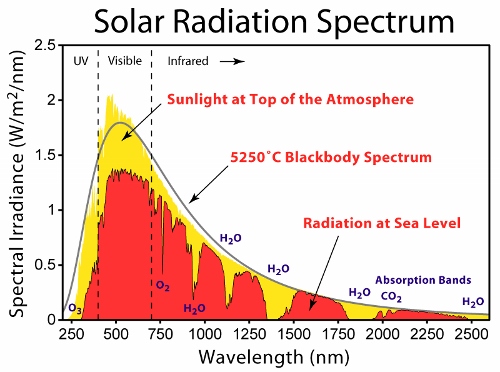
Two papers with a similar theme crossed my social media feeds in the last couple of days. You might think this is just a weird coincidence, but I’m choosing to take it as a sign to write about them for the blog. So, what are these papers, and what’s the theme? One is the final […]